
An interesting correspondence article printed in Lab Animal Europe, Vol 20, July 2020, page 12 entitled “Possible Challenges in behavioral phenotyping of rodents following COVID-19 lockdown” from Kailash Manda should give us all a refreshing look at how we are doing studies and how we are actually getting our data results. She specifically mentions three group factors that affect the data outcomes of behavioral phenotyping (State, Trait and Technical).
We have interviewed Dr John J. Hasenau, one of the most expert Animal Research Veterinarians in Laboratory Animal Science and Principal of Lab Animal Consultants and asked his opinion about this interesting correspondence article.
As we are all aware, John says, many labs have and continue to experience decreased human activities and general city activity levels, and, as mentioned in the article “earth crust vibrations” which can modulate neurobehavioral functions in rodents. Specific “State” factors mentioned were technical skills and experience of the experimenter, Circadian timing, setup and arrangement in the animal room and nesting and bedding surface; while “Trait” factors included, animal handling, stress level of the animal, health and age of the animal, strain and sex of the animal; and Technical was inclusive of, automation of vivarium maintenance, health data record keeping, room macro-environment control and monitoring, and SOPs.
The primary concern, John continues, is ability for reproducibility and repeatability with these activity changes from the COVID-19 responses, and how to understand when this data generated from these animals can be acceptable or used for study analysis, begging the question of what is “normal” rodent behavior for stated strain, health and age of the animals. This correspondence importantly ties a partial answer to baseline data and comparisons but mentions the possibility of transgenerational and intergenerational changes in behavioral functions based on decreased interactions with handling and human interface.
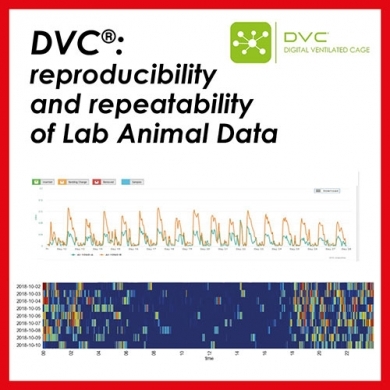
Generating baseline behaviors and getting to normal rodent behaviors depends on behavioral testing apparatus and how mice are handled as mentioned earlier. John underlines that a more recent movement in the industry to decrease variability that can be introduced by frequent and different human handling and interfacing and improving reproducibility and repeatability is an area of automated home cage monitoring1. The automated home cage method of housing animals has contributed to better understandings of “normal” animal activities and behaviors as well as decreasing the need for handling and for human interfacing. It has greatly enhanced our understanding of the circadian timing of strains of mice and a much better understanding of how room size, cage arrangements and human activities affect the activity and behaviors of the animals2. This has also allowed large data accumulations of what is normal for certain strains at various ages and health status 3.
The principal of automated home cage monitoring is to use the rodent home cage for a testing apparatus, decreasing the need for the animals to be handled to be moved to a testing apparatus and to actively capture animals activities and behaviors 24/7 but especially when they are in their highest (nocturnal) activity levels 4,5. This decrease in the human interactions and changing into testing apparatus allow for less stress induction and prolonged acclimations as seen with some studies. It has also allowed better realization of how and when animals are using different areas of a cage defined as zonal components of resting, nesting and elimination behaviors and how these may differ with different cage stocking densities and or animal purposes (i.e. breeding). This type of caging system in this COVID-19 response allows the investigative staff an enhanced certainty of when certain strains of transgenics or knockout rodents are back to normal neurobehavioral baseline levels especially when there has been well developed baseline data for those strains.
One area mentioned in the correspondence, John says, is the ”technological robustness of the animal care and husbandry practices, such as automation in cage changing,” The use of automated home cage monitoring systems align with this statement. One of the systems in use (Digitally Ventilated Caging, DVC®, Tecniplast) allows automation indication of when cages should be changed, which routinely expands out the calendar cage changing time (dependent on cage stocking density and stain usage), decreasing the acoustic and seismic issues associated with cage change outs. This system is uniquely based on Electromagnetic fields and has husbandry management component in addition to the activity monitoring making it more attractive from both a facilities management aspect as well as a study conductance aspect6. In addition the Room Environment Monitoring (REM) system that compliments the DVC allows for evaluation of room activities and influences that occur at the macroenvironmental level that can impact animal activity at the microenvironmental level. This makes the combined DVC and REM systems an extremely robust automated home cage monitoring package allowing both the animal activities and behaviors as well as any precieved room environmental activities (either human, lighting, or HVAC) to be accounted for in data analysis and interpretation.
John concludes that the use of automated home cage monitoring systems during these pandemic times allows greater assurance for the investigator staff to be able to conduct studies with valid neurobehavioral outcomes from the transgenic and knockout animals that have been characterized and studied. This would also be more timely than use of traditional non-home cage monitoring systems due to decreased acclimation times from human interactions.
References:
John J. Hasenau
Laboratory Animal Consultants